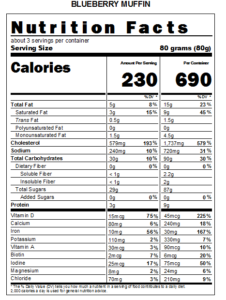
 September 29, 2017 – The United States Food and Drug Administration (FDA) announced they are proposing an extension of compliance dates for the Nutrition Facts and Supplement Facts label final rules of July 26, 2018, to January 1, 2020, for manufacturers that have annual food sales of $10 million or more. The proposed compliance date for manufacturers that have food sales of less than $10 million would be extended to January 1, 2021.
September 29, 2017 – The United States Food and Drug Administration (FDA) announced they are proposing an extension of compliance dates for the Nutrition Facts and Supplement Facts label final rules of July 26, 2018, to January 1, 2020, for manufacturers that have annual food sales of $10 million or more. The proposed compliance date for manufacturers that have food sales of less than $10 million would be extended to January 1, 2021.
There has been increased concerns from stakeholders of food organizations to have their products compliant by the original July, 2018 deadline. A number of technical clarifications need to be obtained from the FDA relating to the final rulings. Most important is to provide accurate information to the consumer so they can make informed decisions about their diet and the foods they consume.
